PentaHibeComplete
Ready-to-use
PentaHibe Complete is a ready-to-use cGMP compliant formulation that can be used with a range of cell types and intestinal mucosal biopsies for organoid generation. With 10% pentaisomaltose and 2% DMSO as cryoprotective agents, PentaHibe Complete significantly reduces DMSO-related toxicity and side effects.
- cGMP manufactured
- Reduced % of DMSO
- Sterile
- Serum-free
- Low endotoxins
- Water-like viscosity
Improved outcomes
PentaHibe Complete, containing 2% DMSO, shows comparable results to commercially available cryosolutions with higher DMSO concentration and even demonstrates superior migratory potential.
Enhanced safety
PentaHibe Complete, containing 2% DMSO, minimizes cellular toxicity and DMSO-related side effects, enhancing both safety and effectiveness vs. traditional DMSO-based cryopreservation methods.
Tested on several cell types
PentaHibe Complete, cGMP manufactured, has been tested on T-cells, human mesenchymal stem cells, and CHO cells and comes with comprehensive documentation to support regulatory submissions.


Data & Results
A ready-to-use cryopreservation medium containing 10% pentaisomaltose and 2% DMSO, PentaHibe Complete can be used used across a range of cell types and for tissue cryopreservation in organoid generation.
T cells

Objectives: to compare and evaluate the viability and recovery of T cells purified from buffy coats after cryopreservation with a self-made 10% DMSO formulation, a commercial product with 10% DMSO, or ready-to-use PentaHibe Complete.
Results: T cells purified from buffy coats from healthy donors and cryopreserved with PentaHibe Complete show similar viability and recovery to those cryopreserved with self-made and commercial formulas containing 10% DMSO.
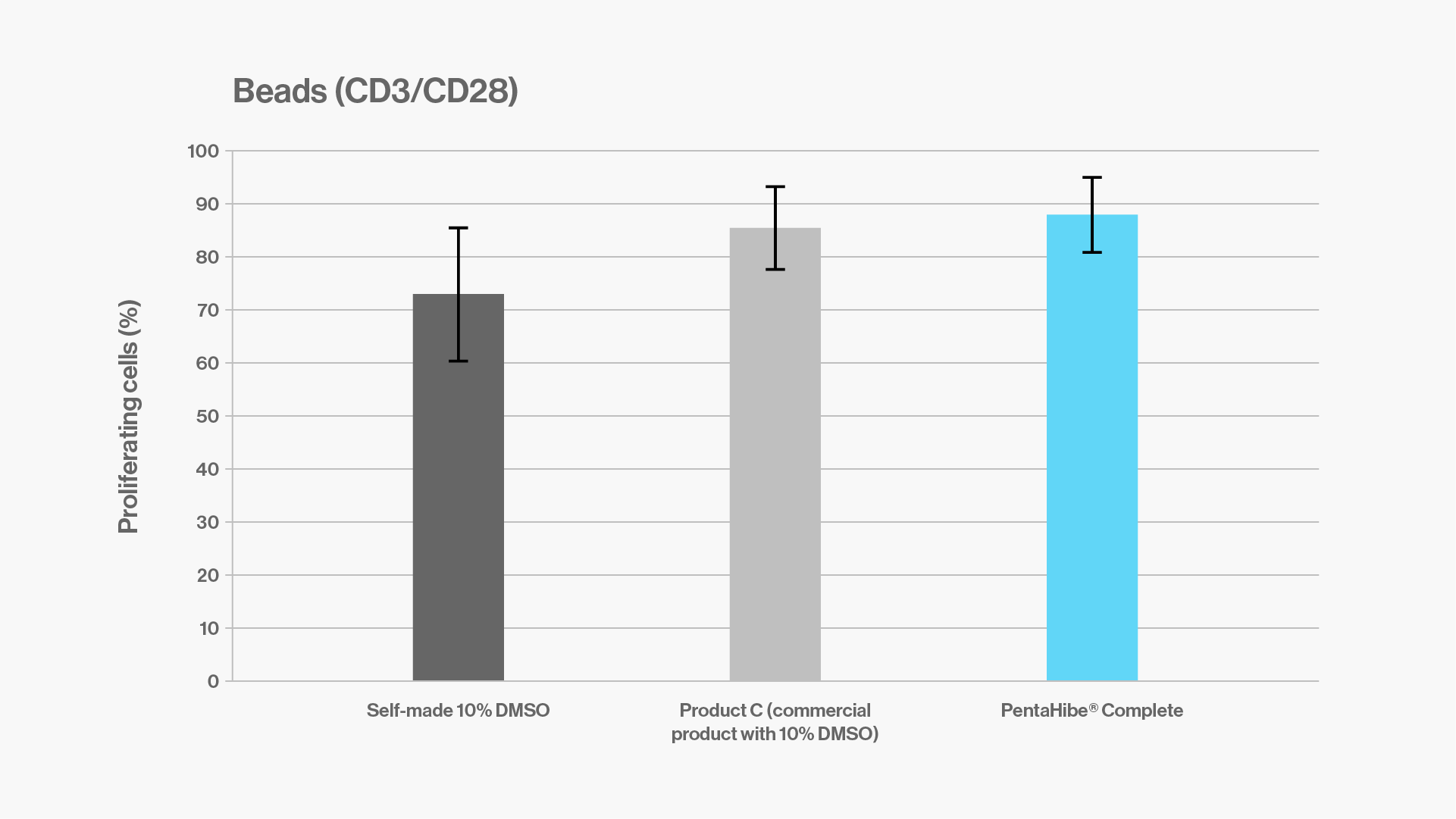
Objectives: to compare and evaluate the proliferation of T cells purified from buffy coats after cryopreservation with a self-made 10% DMSO formulation, a commercial product with 10% DMSO, and ready-to-use PentaHibe Complete.
Results: T cells purified from buffy coats and cryopreserved with PentaHibe Complete show similar proliferation as those cryopreserved with self-made and commercial formulas containing 10% DMSO.

Objectives: to evaluate the expansion capabilities of T cells post-cryopreservation using PentaHibe Complete compared to a commercial product containing 10% DMSO. T cells were stimulated with IL-2 and anti-CD3/CD28 antibody beads, and their expansion was monitored over a 15-day period. Expansions were calculated from the accumulated count of viable T cells on selected days after thawing.
Results: T cells purified from buffy coats of healthy donors and cryopreserved with PentaHibe Complete showed similar expansion comparable to those preserved in a commercial formulation containing 10% DMSO.

Objectives: To evaluate the post-thaw viability of T cells cryopreserved with PentaHibe Complete. Viability was measured at different time points (0, 6, and 24 hours post-thaw). The study aimed to assess the effectiveness of PentaHibe® Complete in maintaining high viability for downstream applications in cell therapy research.
Results: PentaHibe Complete demonstrated exceptional post-thaw viability, with >90% viability immediately after thawing. Over a 24-hour culture period, viability remained high (80-85%), providing an extended window for handling and processing. These findings confirm that PentaHibe Complete is an effective cryoprotectant for preserving the integrity and functionality of T cells in translational research.

Objectives: To evaluate the preservation of post-thaw T cell functionality and effector response following cryopreservation using PentaHibe Complete compared with a commercial cryopreservation product (Product C). Primary human T cells were cryopreserved using a controlled-rate freezing protocol, stimulated immediately after thaw with PMA/ionomycin, and functional activity was assessed by measuring interferon-gamma (IFN-γ) secretion. Values represent mean ± SD (n = 9; 3 donors with triplicate measurements).
Results: T cells cryopreserved in PentaHibe Complete demonstrated strong IFN-γ secretion immediately after thaw, comparable to Product C. These findings show that PentaHibe Complete effectively maintains T cell effector responses following cryopreservation, making it well suited for translational and cell therapy applications.
Human mesenchymal stem cells

Objectives: to compare and evaluate the viability of adipose tissue derived human mesenchymal stem cells after cryopreservation with a self-made 10% DMSO formulation, a commercial product with 10% DMSO, or ready-to-use PentaHibe Complete.
Results: PentaHibe Complete is suitable for the cryopreservation of adipose tissue derived human mesenchymal stem cells.
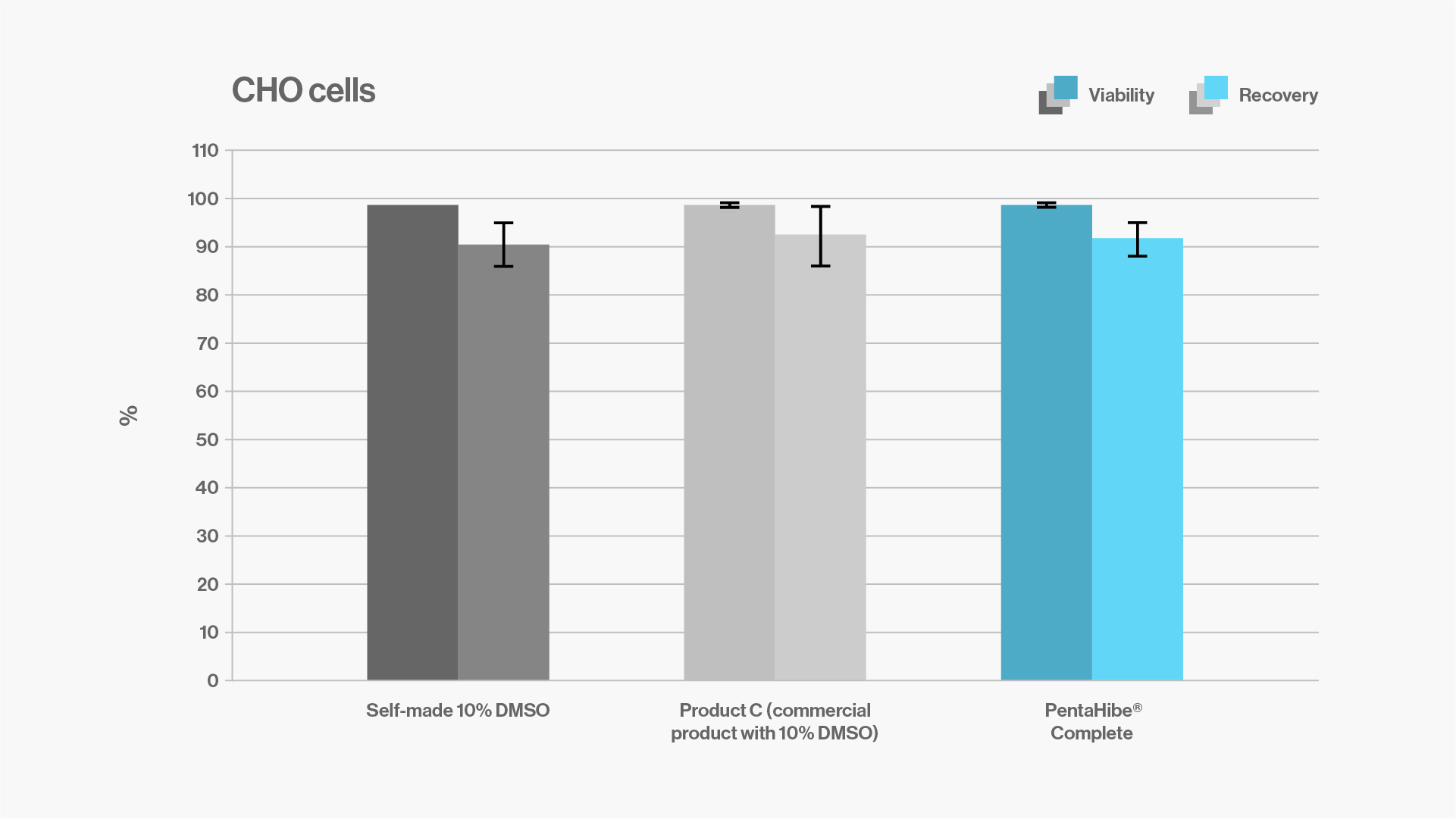
Objectives: to compare and evaluate the viability and recovery of CHO cells after cryopreservation with a self-made 10% DMSO formulation, a commercial product with 10% DMSO, or ready-to-use PentaHibe Complete.
Results: CHO cells cryopreserved with PentaHibe Complete show similar viability and recovery to those cryopreserved with self-made and commercial formulas containing 10% DMSO.

Objectives: to compare and evaluate the viability and recovery of HEK293 cells after cryopreservation with Pentahibe Complete or with a commercial product with 10% DMSO.
Results: HEK cells cryopreserved with PentaHibe Complete show similar viability and recovery to those cryopreserved with commercial formula containing 10% DMSO.
Data generated in the Zilbauer Group, Cambridge Stem Cell Institute

Objectives: to evaluate the ability to successfully establish organoid cultures from intestinal mucosal biopsies (duodenum, ileum, and colon) cryopreserved with either Pentahibe Complete or a conventional 10% DMSO-based medium.
Results: biopsies cryopreserved with Pentahibe Complete demonstrated a 100% organoid establishment success rate, compared to 64.29% for the standard freezing medium, across all intestinal segments.

Objectives: To assess the impact of Pentahibe Complete versus a conventional 10% DMSO-based freezing medium on both the number and area expansion of organoids derived from duodenum (Duo) biopsies over time, using time-lapse imaging analysis.
Results: Biopsies cryopreserved in Pentahibe Complete consistently produced a higher number of organoids and exhibited substantially greater area expansion compared to those frozen in a standard 10% DMSO-based medium over a 180-hour observation period. Time-lapse imaging confirmed these findings, revealing increased organoid count (Fig. 1a) and larger organoid area (Fig. 1b) over time, with representative images demonstrating robust growth by day 13 post-thaw (Fig. 1c). These results underscore Pentahibe Complete as an advanced solution for preserving tissue integrity and promoting efficient organoid generation.⁷
- Pharmacosmos A/S
- Roervangsvej 30
- DK-4300 Holbaek
- Denmark
- Tel: +45 5948 5959
- Email: pentahibe@pharmacosmos.com
- Business hours are Monday to Friday 08:00-16:00 CET
